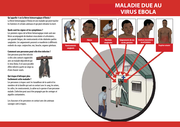
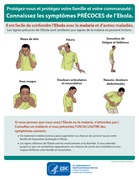
Filter
1866
Text search:
Centers
of
Disease
Control
and
Prevention
CDC
Featured
Recommendations
175
New Publications
516
Language
English
1614
French
139
Spanish
94
Russian
57
Portuguese
36
Arabic
34
Kreyol
19
German
17
Chinese
17
Vietnamese
6
Farsi
5
Indonesian
4
Amharic
4
Turkish
3
Italian
3
Tagalog
2
Urdu
2
Serbian
2
Romanian
2
Bulgarian
2
Greek
2
Croatian
2
Hungarian
2
Polish
2
Ukrainian
2
Hausa
1
Khmer
1
Swahili
1
Bengali
1
Tamil
1
Hindi
1
Malay
1
Thai
1
Korean
1
Punjabi
1
Laotian
1
Bosnian
1
Japanese
1
Hebrew
1
Pashto
1
Document type
No document type
1097
Guidelines
268
Studies & Reports
173
Manuals
93
Strategic & Response Plan
64
Fact sheets
63
Training Material
37
Infographics
21
Situation Updates
17
Resource Platforms
17
Brochures
9
Online Courses
3
Dashboards/Maps
2
Videos
2
Countries / Regions
Sierra Leone
81
Global
76
Liberia
69
Ethiopia
57
Nigeria
53
Kenya
47
India
44
Guinea
42
Congo, Democratic Republic of
40
Senegal
36
Rwanda
34
Uganda
33
Africa
32
Ghana
30
Latin America and the Carribbean
29
Zambia
27
Haiti
26
Tanzania
26
Burkina Faso
25
South Africa
24
Western and Central Europe
24
Côte d’Ivoire / Ivory Coast
20
Mali
19
Namibia
19
Germany
18
Bangladesh
18
Malawi
17
West and Central Africa
17
Brazil
16
Russia
16
USA
15
Cameroon
15
Zimbabwe
14
Chad
13
Mozambique
13
East and Southern Africa
13
Guinea-Bissau
12
Niger
12
Colombia
12
Cambodia
11
Nepal
10
Ukraine
10
Middle East and North Africa
10
Eastern Europe
10
Philippines
9
Indonesia
9
Botswana
9
Madagascar
8
Asia
8
South Sudan
7
Syria
6
South–East Asia Region
6
Vietnam
6
Eswatini/ Swaziland
5
Lesotho
5
Pakistan
4
Ecuador
4
Yemen
4
Paraguay
4
Egypt
3
Thailand
3
China
3
Benin
3
Myanmar / Burma
3
North America
3
France
3
Spain
3
Afghanistan
2
Turkey
2
Western Pacific Region
2
Venezuela
2
Congo-Brazzaville
2
Portugal
2
Saudi Arabia
1
Togo
1
Somalia
1
Lebanon
1
Malaysia
1
Argentina
1
Peru
1
Burundi
1
Honduras
1
Bolivia
1
Angola
1
Cape Verde
1
Mexico
1
Sri Lanka
1
Chile
1
Eastern Europe and Central Asia
1
Bhutan
1
Southern Africa
1
Laos
1
Timor Leste/ East Timor
1
Tajikistan
1
Qatar
1
Kyrgyzstan
1
Kazakhstan
1
Uruguay
1
Japan
1
Belgium
1
United Kingdom
1
South Korea
1
Authors & Publishers
World Health Organization WHO
186
Centers for Disease Control and Prevention CDC
171
World Health Organization
109
et al.
107
World Health Organization (WHO)
58
Centers for Disease Control and Prevention (CDC)
44
USAID
39
Centers for Disease Control and Prevention
36
UNICEF
33
UNAIDS
29
World Health Organisation (WHO)
29
WHO
23
Africa CDC Centres for Disease Control and Prevention
18
African Union
18
CDC
18
Organisation Mondiale de la Santé OMS
17
European Centre for Disease Prevention and Control
15
Ministry of Health, Kenya
15
UNFPA
14
Africa CDC Centers for Disease Control and Prevention
13
PEPFAR
12
National AIDS
11
Ministry of Health and Social Welfare, Tanzania
10
Pan American Health Organisation PAHO
10
Pan American Health Organization PAHO
10
Stop TB Partnership
10
Organisation Mondiale de la Santé
9
CDC Centers for Disease Control and Prevention
8
ICF International
8
Organisation mondiale de la Santé (OMS)
8
Pan American Health Organization
8
Africa Center for Disease Control and Prevention
7
Ministry of health
7
National Center for HIV
7
PLOS One
7
World Health Organization WHO, et al.
7
AIDSinfo
6
Asociación Colombiana de Infectiologiá ACIN
6
Centers for Disease Prevention and Control CDC
6
Ministry of Health and Sanitation Sierra Leone
6
Ministry of Health, Zambia
6
National Department of Health South Africa
6
Organisation Mondiale de la Santé OMS
6
Regional Office for Africa
6
Robert-Koch-Institut RKI
6
United Nations General Assembly
6
United Nations Secretary-General
6
Centers of Disease Control CDC
5
END TB
5
European Centre for Disease Prevention and Control ECDC
5
Fondo de Poblaciones de las Naciones Unidas - UNFPA
5
IACAPAP
5
International Federation of Red Cross And Red Crescent Societies
5
Ministry of Health and Social Services (MOHSS)
5
National AIDS Control Organization (NACO), Ministry of Health and Family Welfare, Government of India
5
Organización Panamericana de la Salud
5
UNHCR
5
United States Agency for International Development
5
UNOPS
5
World Health Organization WHO
5
Africa CDC
4
Centers for Disease Control and Prevention (HHS-CDC)
4
Federal Ministry of Health, Uganda
4
G. Ayala
4
Global AIDS Program (GAP)
4
Government of Tanzania
4
J. Bear
4
J. Butler
4
MEASURE DHS
4
Ministry of Health, Malawi
4
Ministry of Health, Rwanda
4
Pan American Health Organization (PAHO)
4
Pan American Health Organization PAHO, World Health Organization WHO
4
Partnership for Evidence-Based Response to COVID-19 (PERC)
4
UN Children's Fund UNICEF
4
UNDP
4
United States Department of Health and Human Services
4
UpToDate
4
Wolters Kluwer
4
World Health Organization (Americas)
4
World Health Organization WHO, Regional Office of Europe
4
World Health Organization, Regional Office for the Americas
4
Bayerisches Landesamt für Gesundheit und Lebensmittelsicherheit
3
C. Daniels
3
C. Daniels, et al.
3
C. Smyth, et al.
3
Catholic Relief Services CRS
3
CDC United States
3
Center for Excellence in Disaster Management and Humanitarian Assistance
3
Centers for Diseases Control and Prevention
3
Elsevier
3
European Centre for Disease Prevention and Control ECDC
3
European Centre for Disease Prevention and Control (ECDC)
3
Federal Democratic Republic of Ethiopia, Ministry of Health
3
Federal Ministry of Health - Nigeria Centre for Disease Control
3
FG Consulting Group
3
Global Polio Eradication Initiative GPEI
3
Global Task Force on Cholera Control (GTFCC)
3
Health System Trust
3
Human Rights Watch
3
ICAP
3
IFRC
3
International Union Against Tuberculosis and Lung Disease
3
MHSS
3
Ministerio de Salud Pública de Ecuador
3
Ministerio de Salud y Protección Social
3
Ministerio de Salud y Protección Social Bogotá, Colombia
3
Ministère du Plan et Suivi de la Mise en œuvre de la Révolution de la Modernité (MPSMRM), Ministère de la Santé Publique (MSP) and ICF International
3
Ministry of Health and Family Welfare Government of the People's Republic of Bangladesh
3
Ministry of Health and Social Welfare, Liberia
3
Ministry of Health and Social Welfare, National AIDS Control Programme, Tanzania
3
Ministry of Health Uganda
3
Ministry of Health, Federal Democratic Republic of Ethiopia
3
Ministry of Health, Ghana
3
Ministry of Health, Republic of Uganda
3
Ministry of Health, Rwanda & Rwanda Biomedical Centre
3
Ministry of Health, Zambia, et al.
3
NACO
3
National Agency for the Control of AIDS (NACA)
3
Organisation Mondiale de la Santé OMS, et al.
3
PAHO
3
PAHO (Pan American Health Organization
3
President's Emergency Plan for AIDS Relief (PEPFAR)
3
PRESIDENT’S MALARIA INITIATIVE
3
Rev Panam Salud Publica
3
Rwanda Biomedical Center - Institute of HIV
3
United Nations Children’s Fund (UNICEF)
3
University Teaching Hospital,
3
US Committee for Refugees and Immigrants
3
US Dept. of Health and Human Services
3
World Health Organization ( WHO), Regional Office for South-East-Asia
3
World Health Organization (Europe)
3
World Health Organization (WHO), Regional Office for South-East-Asia
3
World Health Organization)
3
Всемирная организация здравоохранения
3
A. Kamarulzaman
2
American Diabetes Association
2
Bousquet, J.
2
Brakarsh, J.
2
British Society for antimicrobial chemotherapy BSAC
2
Brunner B., J. Barnes, A. Carmona et. al.
2
C. Beyrer
2
Center for Disease Control and Prevention CDC
2
Centers for Disease Control and Prevention, National Center for HIV
2
Central Statistical Office Lusaka
2
Christoffel-Blindenmission (CBM)
2
CII Confederation of Indian Industry
2
de Carvalho-Pinto, R. M.
2
Delfini Cançado, J. E.
2
Department of Health and Human Services
2
Department of Health, Republic of South Africa
2
Dr. V. R. Kandula
2
E. Aaraj
2
Engender Health Society (Improving Women's Health Worldwide)
2
Ethiopian Health and Nutrition Research Institute
2
European Centers for Disease Control and Prevention ECDC
2
European Monitoring Centre for Drugs and Drug Addiction
2
F. Saini
2
Federal Ministry of Health Nigeria
2
Federal Ministry of Health, Ethiopia
2
Federal Ministry of Health, Nigeria
2
G. Venugopal
2
Government of India, Ministry of Health and Family Welfare, National AIDS Control Organisation (NACO)
2
Government of Kenya
2
Government of Sierra Leone, Ministry of Health and Sanitation
2
Government of the People's Republic of Bangladesh DGHS Directorate General of Health Services Ministry of Health and Family Welfare
2
H. Getahun
2
ICAP Columbia University
2
Indian Nursing Council
2
INTECH
2
Inter-Agency Standing Committee
2
Inter-Agency Standing Committee IASC
2
International Organization for Migration IOM
2
International Pharmaceutical Federation (FIP)
2
Iris (Institutional Repository)
2
J. Fuentes
2
Johns Hopkins Center for Communication Programs (CCP) - Ethiopia
2
K. Munir
2
Kamps, B.S.
2
Khaltaev, N.
2
L. Steinitz
2
M. Alonso González
2
M. Smelyanskaya, J. Duncan (The Focus Group Consulting)
2
Managment of Health Sciences MSH
2
MEDBOX
2
Menezes Pizzichini, M. M.
2
Ministerio da Saude, Brasilia
2
Ministerio de Salud y Protección Social, Colombia
2
Ministry of Health [Kenya], National AIDS and STI Control Programme (NASCOP)
2
Ministry of Health and Sanitation, Sierra Leone
2
Ministry of Health and Social Services (MOHSS), PEPFAR, Namibia Statistics Agency, Namibia Institute of Pathology Limited
2
Ministry of Health Ethiopia
2
Ministry of Health Ghana
2
Ministry of Health Nepal
2
Ministry of Health Zambia
2
Ministry of Health, Cambodia
2
Ministry of Health, Gabarone, Botswana
2
Ministry of Health, Liberia
2
Ministry of Health, Republic of Botswana
2
MSMGF
2
National Academies of Sciences, Engineering, and Medicine
2
National AIDS Control Organisation (NACO), Ministry of Health & Family Welfare, Government of India
2
National AIDS Control Organisation (NACO), Ministry of Health and Family Welfare (MOHFW), Government of India
2
National Asthma Education Programme
2
National Center for Emerging and Zoonotic Infectious Diseases Division of Vector-Borne Diseases (CDC)
2
National Tuberculosis Control Programme, Directorate General of Health Services, Ministry of Health and Family Welfare, Bangladesh
2
NCD Alliance
2
Nigeria Centre for Disease Control
2
Organisation de la Santé Mondiale
2
Organización Mundial de la Salud
2
OSHA Occupational Safety and Health Administration
2
Pan American Journal of Public Health
2
Pata
2
Philippine Society for Microbiology and Infectious Diseases
2
PMC
2
Population Foundation of India
2
PRONASIDA, et al.
2
Pub Med
2
Public Health Institute
2
ReliefWeb
2
Research Gate
2
Robert-Koch-Institut
2
Routledge
2
S. Seedat
2
SIAPS
2
Smithsonian Science Education Center
2
Smithsonian Science Education Center, et al.
2
T. D. Benton
2
The DHS Program
2
The Ethiopian Public Health Institute, Federal Ministry of Health Ethiopia
2
The World Bank
2
Tropical Diseases Research Centre, Zambia, DHS
2
U.S. Department of Human Services
2
U.S. President's Malaria Initiative (PMI), CDC, USAID, et al.
2
UKAid
2
UNAIDS (Joint United Nations Programme on HIV/AIDS)
2
UNESCO
2
United Nations Office on Drugs and Crime
2
US Agency for International Development
2
USAID, UNICEF, UNFPA et al.
2
WHO, HHS-CDC
2
Word Health Organization
2
World Bank
2
World Health Organization (WHO), National Center on Birth Defects and Developmental Disabilities from the United States Centers for Disease Control and Prevention (CDC), International Clearinghouse for Birth Defects Surveillance and Research (ICBDSR)
2
World Health Organization Europe
2
World Health Organization WHO, Regional Office for Africa
2
World Health Organization, Regional Office for Africa
2
World Health Organization, UN High Commissioner for Refugees
2
Министерство здравоохранения Российской Федерации
2
(CDC) Center for disease control and prevention
1
, S. Eymard-Duvernay
1
4 Children
1
[Global Handwashing Partnership]
1
A. Rimoin
1
A. A. Neilson, C. A. Mayer
1
A. Abdul-Quader
1
A. Abdul-Quader, M. Berry, T. Bingham
1
A. Armstrong
1
A. Baddeley
1
A. Baller, et al.
1
A. Bolotbaeva
1
A. Boltaev
1
A. Cournil
1
A. E. Kendall
1
A. Haenen
1
A. Jindani, A. Nunn, D. A. Enarson
1
A. Lachman
1
A. Mahieu
1
A. Malaza
1
A. Rakotoarisoa, L. Randrianasolo, S. Tempia, et al.
1
A. Spaulding
1
A. Turkova
1
A. van Ahee
1
A. Wirtz
1
A. Yager
1
A.Lachman
1
A.Ramesh
1
A.Sullivan
1
Aboud S., R. Mdegela
1
Abras A., Ballart C., Fernández-Arévalo A., et al.
1
Academy for Educational Development
1
ACC
1
Access to Medicine Foundation
1
achieving control of asthma in children in africa - acacia
1
Adams, J.
1
adapted from Bang et al., Ann Intern Med (2009)
1
Aday A.W.
1
Addis Ababa City Administration Health Bureau
1
Addiss, D.
1
Adriana Jiménez Rojas
1
AECT
1
Africa Region
1
African Palliative Care Association
1
AIDS
1
AIDS & TB Programme
1
AIDS and Behavior
1
Alemu Tilahun, H.
1
Alexander Butchart, Susan Hillis, Angela Burton et al.
1
Alice Mark
1
Alice Payne Merritt, Gabrielle Hunter, Anne Ballard et al.
1
Allers, Claudia, Timothy O’Hearn, and Meba Kagone
1
Alliance for Public Health
1
ALNAP
1
Alonso González, Mónica
1
Alzheimer's Association
1
Amaya M Gillespie, Rafael Obregon, Rania El Asawi, et al.
1
AmC
1
American College of Cardiology - NCD Academy
1
American Diabetes Association in collaboration with the Centers for Disease Control and Prevention (CDC)
1
American Public Health Association
1
AMREF
1
Amrita Gill-Bailey, Kathryn Bertram, Uttara Bharath et al.
1
Amy Bloom, Leo Blanc, Chris Dye et al.
1
Ana Carvajal, José Félix Oletta López, Alejandro Rísquez
1
and A. Kent
1
and C. Hoffmann
1
and P. Reijer
1
and S.J. Mason
1
Andrade, D.
1
Anne Griffin, Patricia A. Potter
1
Ansari, Armin
1
Antares Foundation
1
Arakaki-Sanchez
1
Arbeitskreises Blut des Bundesministeriums für Gesundheit
1
Ardath Grills, Stephanie Morrison, Bradley Nelson et al.
1
Arias, Daniel
1
Arlington, VA: USAID’s AIDS Support and Technical Assistance Resources, AIDSTAR-One, Task Order 1
1
Armed Forces Pest Management Board, USA
1
Asia Pacific Observatory on Health Systems and Policies
1
Asian-Pacific Resource & Research C0entre for Women (ARROW)
1
ASLM
1
Aten, J.
1
August Burns, Linda O’Neal Eckert, Susan Hariri et al
1
Australian Commission on Safety and Quality in Health Care (ACSQHC)
1
Awudu, RA.
1
Ayalew, A. et al.
1
B. Aracena-Genao
1
B. B. Bachore
1
B. C. Zanoni
1
B. Diaz
1
B. Herstad
1
B. James, Ph.D.
1
B. Karo
1
B. Moges, B. Amare, F. Asfaw, et al.
1
B. Pick
1
B. Rajamaki
1
B. Weil
1
Balaji, V., P. Rupali, K. Walia, et al.
1
Barben et al.
1
Bardosh, K.
1
Barker, G.
1
Barnes, H.
1
Bartlett, E. S.
1
BBC Media Action
1
Beard, R. Suzanne
1
Becher, H.
1
Belgian Antibiotic Policy Coordination Committee
1
Benjamin J. Park, MD
1
Benjamin O Alli [et al.]
1
Berlin, E.
1
Bernard M. Branson, S. Michele Owen, Laura G. Wesolowski et
1
Berry, P.
1
Bham A., J. Bhiman, F. Bongweni et al.
1
Bham, A.
1
Bill & Melinda Gates Foundation
1
Bill & Melinda Gates Foundation, GAVI, et al.
1
Bio Med Central
1
BMC Infectious Diseases
1
BMC Public Health (BioMed Central)
1
BMJ Global Health
1
BMJ Journals
1
BMJ PACK
1
Boeuf, P.
1
Bonnie L. Shepard
1
Bouman, D., W. Novalia, P. Hiemstra, et al.
1
Bower, J.
1
British Medical Journal
1
BRT Business Responds to AIDS (Stopping AIDS is everyone's Business)
1
Bruce Cogill
1
Bulletin of the World Health Organization
1
Bush, A.
1
C. Contini, M. Maritari, M. die Nuzzo, et al.
1
C. Cravero, S. Danner et. al.
1
C. Fitzpatrick, J.Jannin
1
C. Harrison et al.
1
C. Hayashi
1
C. Hoffmann und J. K. Rockstroh
1
C. J. Berg
1
C. Kollan
1
C. L. Moore
1
C. M. Yuen, H. O. Weyenga, A. A. Kim, et al.
1
C. R. Vitek
1
C. Rae
1
Camp, R. and C. Hoffmann
1
Cancer Association of South Africa (CANSA)
1
Carla Silva-Matos
1
Casa
1
Caspary, Kevin
1
Castro,, P.
1
Catholic Relief Services
1
CDA Collaborative Learning Projects
1
CDC Center for Disease Control and Prevention (U.S. Department of Health and Human Services)
1
CDC et al.
1
CDS
1
Cecchini, M.
1
cedpa (The Centre for Development and Population Activities)
1
Center for Disease Control
1
Center for Disease Control (CDC), Climate and Health Program
1
Center for Disease Control and Prevention
1
Center for Disease Control and Prevention (CDC)
1
Center for Disease Control and Prevention, World Health Organisation
1
Center for Operational Analysis and Research COAR
1
Centers for Disease C ontrol and Prevention (CDC )
1
Centers for Disease Control & Prevention
1
Centers for Disease Control and Prevention (CDC)
1
Centers for Disease Control and Prevention (CDC), American Water Works Association
1
Centers for Disease Control and Prevention , World Health Organization WHO
1
Centers for Disease Control and Prevention CDC, American Nurses Association ANA
1
Centers for Disease Prevention and Control
1
Centers of Disease Control and Prevention CDC
1
Central Statistical Agency [Ethiopia] and ICF International
1
Central Statistical Office (CSO) [Swaziland], and Macro International Inc
1
Central Statistical Office (CSO) Zambia, Ministry of Health (MOH) Zambia, and ICF, et al.
1
Central Statistical Office (CSO), Ministry of Health (MOH), and ICF International
1
Central Statistical Office (CSO), Ministry of Health, University Teaching Hospital (UTH)-Virology Laboratory, Tropical Diseases Research Centre (TDRC), Department of Population Studies at the University of Zambia (UNZA)
1
Central TB Divison, Ministry of Health and Social Welfare, Government of India
1
Centre de collaboration nationale sur les politiques publiques et la santé
1
Centre d’études stratégiques de l’Afrique
1
Centre for Respiratory Diseases and Meningitis and Outbreak Response
1
Centro de Coordinación de Alertas y Emergencias Sanitarias
1
CERAH
1
Chagas Coalition
1
Challenge TB
1
Child Health Division
1
Children & AIDS
1
Cholera Secretariat Zimbabwe
1
Chowell and Nishiura
1
Christina Brandes-Barbier, Véronique Millot, Tomas Allen et al
1
Cilliers, J.
1
Circulation Research
1
Clinical Care Options
1
Clinical Infectious Diseases
1
Clinical Info HIV
1
Clinical Info HIV gov.
1
Clinical Microbiology Reviews
1
ClinMed International Library
1
Cobas
1
COC Netherlands and Columbia University – ICAP (South Africa)
1
CollSeck,A.M.
1
Colombia. Ministerio de Salud y Protección Social
1
Communicable Disease Control Branch - Manitoba Health
1
Communication for Devleopment4 Network
1
Community Health Services Unit, Ministry of Health, Department of Nutrition, HIV and AIDS Lilongwe, Malawi
1
Compaign for tobacco-free Kids
1
Conflict and Health
1
Congressional Research Service
1
Connolly, M.A.
1
Conselho Nacional de Combate ao HIV e SIDA (CNCS), Joint United Nations Programme on HIV and AIDS (UNAIDS)
1
Constella Futures
1
Cooper et al. BMC Medicine
1
Corfmat, J.
1
COU Family TV
1
Craig, J.
1
Crofts, K.
1
Culture and Development Section
1
Cureus
1
Curry International Tuberculosis Center, State of California, California Health & Human Service Agency, Department of Public Health
1
D.
1
D. Cohen
1
D. H. Mulligan,
1
D. Kerrigan
1
D. Lowe, D. Hales
1
D. O. Frauches
1
D. Sculier
1
D. Stewart
1
D. Wolfe
1
Dan Hanfling, John Hick, and Clare Stroud
1
Danel, Isabella
1
Danish Red Cross
1
Danyuttapolchai, J., S. Kittimunkong, S. Nateniyom, et al.
1
David Beran
1
Deininger, K.
1
Delphine Sculier, Haileyesus Getahun, Reuben Granich
1
Dennis Falzon, Elizabeth Harausz, Ernesto Jaramillo et al.
1
Dennis Warner, and Christopher Seremet
1
Department of Health & Human Services USA
1
Department of Health (DOH)
1
Department of Health and Human Services - USA
1
Department of Planning and Health Information, Ministry of Health Cambodia
1
Dept. of Psychiatry
1
Deutschen Gesellschaft für Pädiatrische Infektiologie (DGPI) e.V.
1
Development of Health and Human Services USA
1
DGVS
1
Dheda, K.
1
DHS Program
1
Diallo A. B., A. I. Kollo, M. Camara et. al.
1
Differentiated service delivery
1
Dione D., L. Toure, G. Maynard-Tucker
1
Direction de Lutte contre la Maladie, Ministère de la Santé Publique, République Démocratique du Congo
1
Directorate General of Health Services , Ministry of Health & Family Welfare, Government of India
1
Division of Reproductive Health, National Center for Chronic Disease Prevention and Health Promotion, Coordinating Center for Health Promotion, Centers for Disease Control and Prevention, Department of Health and Human Services,
1
Doyle, M.P.
1
Dr. F. Amond
1
Dr. Joseph Sitienei OGW
1
Dr. M. Dara
1
Dr. Md. R. Karim
1
Dr. N. Mohammad
1
Dr. P. Amoth
1
Dr. S. K. Kibias
1
Dressler D. D., MD, MSc Director OF
1
drra
1
Dutra, W.
1
D’A. Richardson
1
D’Arcy Richardson, PATH, and Greg Pirio, Empower Communications
1
E Clinical Medecine (Published by the Lancet)
1
E. Abrams
1
E. André
1
E. Heseltine
1
E. Korenromp
1
E. McCloskey
1
E. Okitolonda
1
E. Serván-Mori
1
E.G. Brown Jr, D.S. Dooley, K. Smith
1
EACS European AIDS Clinical Society (Европейского клинического общества СПИДа)
1
Education International
1
Edward O'Neil Jr
1
Elaine Abrams, Wafaa El-Sadr, and Miriam Rabkin
1
Eliud Wandwalo, Claire Moodie, Yared Kebede Haile, Amos Kutwa, Odile Ferroussier, Chawalit Natpratan
1
Elly T. Katabira,Moses R. Kamya, Israel Kalyesubula, Alice Namale
1
Elsevir
1
Emergency Medical Services Authority (EMSA) California
1
Emmanuel O. Irek, Adewale A. Amupitan, Temitope O. Obadare, Aaron O. Aboderin
1
EMTCT Global Validation Advisory Committee, Global HIV, Hepatitis and Sexually Transmitted Infections Programmes
1
EngenderHealt;
1
EngenderHealth
1
Epilepsy Foundation
1
Epilepsy Foundation of America
1
Erica Balligand, Michiel Costers and Evelyne Van Gastel
1
Erin Berry-Bibee, Melissa Chen, Kathryn Curtis et al.
1
et al. (Джеймса Баера
1
et al.)
1
et. al
1
et. al.
1
etc al.
1
Ethiopia Central Statistical Agency and ICF International
1
EuroHealth Observer
1
European Medicines Agency EMA
1
European Network for Diagnostics of Imported Viral Diseases
1
European Observatory on Health Systems and Policies Eurostation
1
European specialist nurse organisation (ESN)
1
Eurosurveillance (Europe’s journal on infectious disease epidemiology, prevention and control)
1
Ewig S.
1
Extending Service Delivery (ESD) Project, USAID
1
Ezea, CO.
1
Ezewudo, BI.
1
F. Dockhorn Costa Johansen
1
F. Fezeu
1
F. Gebissa
1
F. O. Fasina, Fasanmi O.G., Makonnen Y. J., et al.
1
F. Pérez
1
F. Samuels
1
F. Stuer
1
F. Tafese
1
Family Health International (FHI)
1
Fanjul, G.
1
FAO
1
FAO, OIE, WHO
1
Fast, Larissa and Adele Waugaman
1
Federal Bureau of Prisons
1
Federal Democratic Republic of Ethiopia -Ministry of Health
1
Federal Government of Nigeria
1
Federal HIV
1
Federal Ministries of Agriculture, Environment and Health
1
Federal Ministry of Health Nigeria , World Health Organisation (WHO)
1
Federal Ministry of Health of Ethiopia
1
Federal Ministry of Health, Ethiopia
1
Federal Ministry of Health, Dept. of Public Health, National HIV
1
Federal Ministry of Health, Federal Republic of Nigeria
1
Federal Ministry of Health, Hygiene and Environment Health Department Ethiopia
1
Feiterna- Sperling, C., F. Brinkmann, C. Adamzik, et al.
1
Fédération Internationale des Societés de la Croix-Rouge et du Croissant Rouge
1
FHI 360
1
fhi360
1
FIND
1
Fistula Care
1
Fitzgerald, D.A. et al.
1
Fleisher, Lisa
1
Flor, L.S.
1
Flying Publisher, Boehringer Ingelheim
1
Focus 1000
1
Fonds des Nations Unies pour la population
1
Food and Drug Administration (FDA)
1
Food and Nutrition Technical Assistance Project
1
Food for the Hungry et al.
1
Francis Aboagye-Nyame, Heather Alexander, Zelalem Gizachew Belachew et al.
1
Frontiers in Pediatrics
1
Fuentes J, Bakare M, Munir K et al.
1
Fundo de População das Nações Unidas
1
G, Zambrano, E Salgato, F. M. Moyano et al.
1
G. F. Tchouwa
1
G. H. Loneragan, H. M. Scott et al.
1
G. H. Mazurek, J. Jereb, P. LoBue et. al
1
G. Jonboboeva
1
G. K. Siberry
1
G. M. Montes Zuluaga, F.N. Alcalde-Rabanal et al
1
G. Pirio
1
G. Ravasi
1
G.S. Wallace& J.F. Seward
1
Gadea, P.
1
Ganina L.Yu
1
GBI
1
Gebreselassie, Tesfayi, and Pav Govindasamy
1
Geffert,K., N. Rupp und S. Mauder
1
Georgios Nikolopoulos, University of Cyprus
1
Gerald Keusch, Keith McAdam, Patricia Cuff, Michelle Mancher, and Emily R. Busta
1
Germany’s National Public Health Institute (RKI), et al.
1
Ghana AIDS Commission
1
Ghana Health Service
1
Ghana Health Service (GHS)
1
Ghana Statistical Service
1
Gibson, A.
1
Gilma Mantilla
1
Giovanny Vinícius Araújo de França, Valdelaine Etelvina Miranda de Araújo et al.
1
Global Antibiotic Resistance Partnership—Tanzania Working Group
1
Global Fund
1
Global Initiative for Asthma
1
Global Laboratory Initiative (GLI), Stop TB Partnership
1
Global Partners in Care
1
Global Polio Eradication Initiative
1
Global Polio Eradication Intiative
1
GOARN
1
GOARN Risk Communication and Community Engagement (RCCE) Coordination Working Group, et al.
1
GOARN, FAO, et al.
1
Goff, D.
1
Goh, N. S. L.
1
Golden, A.
1
Gollob, K.
1
Government of Ethiopia
1
Government of India, Ministry of Health & Family Welfare, Department of Health & Family Welfare
1
Government of Liberia
1
Government of Malawi, Ministry of Health
1
Government of Nepal, Ministry of Health and Population, National Centre for AIDS and STD Control
1
GOVERNMENT OF SIERRA LEONE MINISTRY OF HEALTH AND SANITATION
1
Government of Sierra Leone, Ministry of Health and Sanitation Sierra Leone
1
Government of Sierra Leone, Ministry of Health and Sanitation, Health Education Division
1
Government of the Democratic Republic of the Congo, World Health Organization
1
Government of the People’s Republic of Bangladesh, Directorate General of Health Services
1
Government of the People’s Republic of Bangladesh, Ministry of Health and Family Welfare, Directorate General of Health Services
1
Government of the Republic of Namibia
1
Government of the Republic of Zambia
1
Government of Uganda
1
Governments of Guinea, Liberia, Sierra Leone
1
GP clinics
1
Green Ink
1
Green, R.
1
Gromek K., Hung M., Montero N. et al
1
H. Becher
1
H. Drobna, C. Mallouris
1
H. Flynn
1
H. Schünemann, S.T. Aung, F. Bonsu et al.
1
H.E. Drummer
1
Haitham Shoman, Emilie Karafillakis and Salman Rawaf
1
Halaharvi, H.
1
Haldane, V., De Foo, C., Abdalla, S.M. et al.
1
Harvard Global Health Institute
1
Haut Conseil de la Santé Publique
1
Health Action in Crisis
1
Health Communication Capacity Collaborative
1
Health policy and planning
1
Health Systems and Policy Research
1
HEAT, UNICEF, Open University, AMREF
1
Henry B. Perry, Ranu S. Dhillon, Anne Liu, et al.
1
HHS Panels (*)
1
HIV law project
1
Honein MA, Christie A, Rose DA, et al.
1
Hopkins, D.
1
Hossain, I.
1
Hoy, R.
1
HRSA
1
Hua Fu
1
Human Resources for Health
1
Humanitarian Disaster Institute
1
I. J. C. Schneider
1
I. Scharuk, O. Leonenko-Brodetskaya, O. Sidorenko, et al.
1
I. Semini
1
I. Steffens
1
I. T. Katz
1
IASC et al.
1
IATT
1
ICAP Columbia University (Mailman School of Public Health)
1
ICF International, et al.
1
ICF Macro, USA
1
ICRC
1
ICRW ( International Center for Research on Women)
1
ICW Global (The International Community of Women Living with HIV/AIDS)
1
Idika, IK.
1
Idro R, Musubire K A, Byamah Mutamba B
1
iMedPub
1
Indian Council of Medical Research
1
Indian Council of Medical Research (ICMR)
1
Indonesian Heart Association
1
Indonesian National AIDS Commission
1
Indonesian Urological Association
1
Inés Garcia Baena, Andrew Siroka, Amy Collins et al.
1
Infections & Morbidities
1
Institut National de la Statistique (INS) [Rwanda], Ministère de la Santé (MINISANTÉ) [Rwanda] et Macro International Inc
1
Institut National de la Statistique et de la Démographie Ouagadougou, Burkina Faso
1
Institute for Community Inclusion at University of Massachusetts
1
Institute for Health Metrics and Evaluation (IHME)
1
Institute for Security Studies ISS
1
Institutional Repository for Information Sharing iris
1
Institutional research information sharing iris
1
Inter-Agency Task Team to Address HIV in Humanitarian Emergencies
1
Inter-Agency Task Team to Adress HIV in Humanitarian Emergencies
1
Interagency Task Team HIV in Humanitarian Emergencies
1
International AIDS Society (IAS)
1
International Association for Child and Adolescent Psychiatry and Allied Professions
1
International Bank for Reconstruction and Development/ The World Bank
1
International Diabetes Federation
1
International Federation of Red Cross And Red Crescent Societies IFRC
1
International Medical Corps
1
International Oranization for Migration
1
International Pharmaceutical Federation FIP
1
International Rescue Committee
1
International Research Institute for Climate and Society
1
International Society of Nephrology
1
International Union Against Tuberculosis and Lung Disease, World Health Organization
1
International Vaccines Task Force (IVTF)
1
IntraHealth International
1
IOM UN Migration
1
IPAS
1
IPPF ESEAOR, UNFPA
1
IPPF, UNFPA, WHO, UNAIDS
1
Ipsos
1
Irish Aid
1
Istivto Svpreriore di Santità
1
J. Atkinson, Y. Chartier, C.L.Pessoa-Silva, et.al
1
J. Baer
1
J. Bhiman, F. Bongweni et al.
1
J. Buntine
1
J. Burnett
1
J. Dee
1
J. E. Haberer
1
J. Gao
1
J. Gaugler, Ph.D.
1
J. Hourcade Bellocq
1
J. Kahn, A.Mastroianni, and L.S. Parker
1
J. King
1
J. Langer and J. Slawomirski
1
J. Liu
1
J. M. Garcia Calleja
1
J. M. Rey
1
J. M. Scofield et al.
1
J. Noeske
1
J. Painter, P. Douglas, et al.
1
J. Peter Figueroa
1
J. Smith
1
J. V. Ershova, G. V. Volchenkov, D. A. Kaminski, et al.
1
J. Vujcic, L.Blum, and P.K.Ram
1
J. Wasserheit, H. Schünemann, et cl.
1
J.-I. Cakalo
1
J.M.Mangan, Z. Tillova, et al.
1
J.S. Richards, et al.
1
J.Smith
1
JAMES MACHARIA
1
Janet Ninnis, Ronald S. Cohen, Adam Czynski et al.
1
Japan international cooperation agency JICA
1
Jassat, W.
1
Jean-Pierre Coulaud, George Schmid, ИгорьТоскин et al.
1
Jennings, B.
1
Jilian A Sacks, Elizabeth Zehe, Cindil Redick et al.
1
John Hopkins Bloomberg School
1
John Hopkins Bloomberg School of Public Health
1
John Kinsman
1
Johns Hopkins University and US Agency for International Development (USAID)
1
Joint HIV, TB and PMTCT Review Steering committee
1
Joshi, M.P.
1
Journal of the International AIDS Society
1
Julián-Jiménez, A.
1
Junitos pelo fin da Tuberculose
1
Justice and Corrections Service
1
K. Holland, B. Bartholow, et al.
1
K. Asedeu
1
K. Burns
1
K. Butler
1
K. Ciceri
1
K. Cranston, A. DeMaria (Bureau of Infectious Disease)
1
K. Geffert und S. Mauder
1
K. Greenaway
1
K. Hagmaier
1
K. Hiban, I. Frost, et al.
1
K. Mangino
1
K. Mason
1
K. Wilson
1
Kanu, H.
1
Kasipo, Charles
1
Kato-Wallace, J.
1
Kazembe, L.N. & Neema, I.
1
Kelsey Simmons, Zuki Mihyo and Lyn Messner
1
Kendall, Tamil
1
Kenya Medical Research Institute
1
Kenya Ministry of Health
1
Kenya MInistry of Health, Community Development, Elderly and Children
1
Kenya Ministry of Public Health and Sanitation
1
Kenya National Bureau of Statistics, Ministry of Health, National AIDS Control Council, Kenya Medical Research Institute, National Council for Population and Development, Kenya,The DHS Program, ICF International
1
Khalil, A.
1
Klinkenberg, E
1
KNCV TUBERCULOSIS FOUNDATION
1
KNV
1
Kokki, M.
1
Kompetenznetz Pulic Health Covid-19
1
Konrad-Adenauer- Stiftung
1
Kontsevaya A, Farrington J, Balcılar M, Ergüder T
1
Koren, H.
1
Krause G., M. Borchert, J. Benzler et al.
1
Kumar Roy., S. and S. Bhattacharjee
1
KZN Provincial ARV
1
L. Cai
1
L. Newman
1
L. Rumble, A. A. Ramly, M. Nuryana, et al.
1
L. Telisinghe, K. L. Fielding, J. L. Malden, et al.
1
L.E. Escobar, R. Lowe et al.
1
Laaser, U., F. Beluli
1
Lamunu M, Lutwama J.J, Kamugisha J
1
Lancet Migration
1
LaRocque, R.
1
Laurence Cibrelus
1
Lawrence, E.
1
LeBeau, D. & Yoder, P.S.
1
Lenney, W.
1
Leong, T. L.
1
Liberia Institute of Statistics and Geo-Information Services (LISGIS) Monrovia, Liberia
1
Linda Rudolf, Julian Caplan et al
1
Linda Young Landesman,
1
Llyod, A.
1
Lode, H.
1
Loma Linda University
1
LSHTM
1
Lukito, A.A., A.U. Rahajoe, L.I. Rilantono, et al.,
1
Lungenliga Schweiz, Bundesamt für Gesundheit
1
M. A. Pallansch
1
M. Allen
1
M. Allison Arwady, Luke Bawo, Jennifer C. Hunter et al
1
M. Archary
1
M. B. Mello
1
M. Bakare
1
M. Berry
1
M. Beusenberg et al.
1
M. Botticelli (Bureau of Substance Abuse Services)
1
M. Bray, M.S.Hirsch, D.Mitty, et al.
1
M. Camacho
1
M. Dallao
1
M. Dara, C. D. Acosta, V. Rusovich, et al.
1
M. Dara, M. Grzemska, M. E. Kimerling, et al.
1
M. Djibuti
1
M. F. Biudes
1
M. Hoenigl
1
M. J. Abzug
1
M. J. Stirratt
1
M. Janier, V. Hegyi, N. Dupin et al
1
M. Mansfeld
1
M. Mikulencak
1
M. Outeda
1
M. Rose
1
M. Smelyanskaya, J. Duncan
1
M. Sundararaj
1
M. Wareham
1
M.Bakare
1
M.J. Choi
1
Machalaba, C.
1
Madhukar Pai
1
Maisha (National AIDS Control Council)
1
Marchissio, E.
1
Maria Virginias Villegas
1
Marie Stopes Uganda, WHO
1
Mark F. Cotton
1
Mark, A.
1
Martinez, E.
1
Martini M, M. Abdiker
1
Marty, F.M.
1
Mary Kay Kindhauser, Tomas Allen, Veronika Frank, et al.
1
Mary Leakey et al.
1
Massachusetts Department of Public Health
1
Maternal and Child Health DirectorateFederal, Ministry of Health Ethiopia
1
Mathieu Maheu-Giroux, Lawrence Joseph, Patrick Belisle and others
1
Matsushita, K.
1
Mauder, S.
1
May L., K. Yadav, S. D. Gaona
1
Measles Initiative
1
Medizin Fokus Verlag
1
Meershoek A, Zwerling A, Daftary A, Citro B, Smyth C, Lewis D
1
Mehtsun, Sinit
1
Mental Health and Psychosocial Support Network
1
Médecins sans Frontières
1
Médecins sans Frontières (MSF)
1
mHealth Kenya
1
MHPSS.net
1
Michael T. Osterholma, Kristine A. Moorea, Nicholas S. Kelley et al.
1
Migration Policy Institute
1
Military Medical Research
1
Miller, C.
1
Ministerio da Saude Brasilia
1
Ministerio de Salud Chile
1
Ministerio de Salud Publica y Bienestar Social
1
Ministerio de Salud Pública y Bienestar Social Paraguay
1
Ministerio de salud y deportes, Bolivia
1
Ministerio de Salud, Peru
1
Ministerio de sanidad - España
1
Ministère de la Santé et de l’Action Sociale et Ministère de la Justice
1
Ministère de la Santé et l'Action Sociale, Senegal
1
Ministère du Plan et Macro International. 20
1
Ministério da saúde
1
Ministério da Saúde (MISAU) e Institudo Nacional de Estatística (INE)
1
Ministério da Saúde Direcção Nacional de Saúde Pública Programa Nacional de Controlo da Tuberculose
1
Ministério da Saúde, Brasil
1
Ministério da Saúde. Secretaria de Vigilância em Saúde, Brasil
1
Ministry of Health & Child Welfare
1
Ministry of Health & Family Welfare, India
1
Ministry of Health & Family Welfare, India, et al.
1
Ministry of Health & Sanitation, Sierra Leone
1
Ministry of Health (MoH), Lesotho and ICF International
1
Ministry of Health (MoH), Malawi and ICF International
1
Ministry of Health and Child Care, Zimbabwe
1
Ministry of Health and Sanitation (Sierra Leone), UNICEF, Helen Keller International, and WHO
1
Ministry of Health and Sanitation Sierra Leone,
1
Ministry of Health and Social Development of the Republic of Kazakhstan Republican Center for AIDS Prevention and Control
1
Ministry of Health and Social Services (MOHSS) , PEPFAR, Namibia Statistics Agency, Namibia Institute of Pathology Limited
1
Ministry of Health and Social Services (MoHSS), Namibia
1
Ministry of Health and Social Services (MOHSS), UNAIDS
1
Ministry of Health and Social Welfare (MoHSS), Kingdom of Lesotho
1
Ministry of Health and Social Welfare, The United Republic of Tanzania
1
Ministry of Health Bangladesh
1
Ministry of Health DR Congo
1
Ministry of Health Republic of Ghana
1
Ministry of Health Rwanda, Biomedical Center Institute of HIV
1
Ministry of Health Rwanda, The DHS Program, et al.
1
Ministry of Health Saudi Arabia
1
Ministry of Health Uganda, WHO, UBOS, CDC
1
Ministry of Health, Central Statistics Office, PATH Malaria Control and Elimination Partnership in Africa, et al.
1
Ministry of Health, Ethiopia
1
Ministry of Health, Gaborone, Botswana
1
Ministry of Health, Ghana, World Health Organizaiotn (WHO) and Ghana Health Service
1
Ministry of Health, Government of Liberia
1
Ministry of Health, Haiti
1
Ministry of Health, Liberia, World Health Organization (WHO), and Centers for Disease Control and Prevention (CDC)
1
Ministry of Health, National AIDS and STI Control Program (NASCOP)
1
Ministry of Health, Republic of Kenya
1
Ministry of Health, South Africa
1
Ministry of Health, Swaziland
1
Ministry of Health, Tanzania
1
Ministry of Health, Uganda
1
Ministry of Health, World Health Organisation (WHO), Roll Back Malaria
1
Ministry of Public Health, Quatar
1
Ministry of State for Special Programmes (MSSP) and Ministry of Provincial Administration and Internal Security - National Disaster Operation Centre (NDOC), Kenya
1
Ministry pf Health and Sanitation Sierra Leone
1
Mission Rabies
1
Mohammed A. , Putnis N. et al.
1
Morbidity and Mortality Weekly Report
1
Morbidity and Mortality Weekly Report MMWR
1
Moriana, S.
1
Mukhtar A Adeiza, Abdullah A Abba, Juliana U Okpapi
1
Muñiz F. J., de Casado G. C. , et al.
1
Murni I. K., T. Duke, S. Kinney et al.
1
Murray, C.A.
1
N. B. Karkara
1
N. Bolokadze, et al.
1
N. Chkhartishvili
1
N. Green
1
N. Mdaka
1
N. Nguyen
1
NACO, DOTS
1
Nagel, F.
1
NAIIS Nigeria
1
Naik, W. & Kaneda, T.
1
Nam Aidsmap
1
NASCOP (National AIDS & STI Control Programme), Kenya
1
NASTAD (National Alliance of State & Territorial AIDS Directors)
1
NASTAD Zambia
1
National Academy of Medicine
1
National Agency for the Control of AIDS (NACA), Abuja Nigeria
1
NATIONAL AIDS and STIs CONTROL PROGRAMME (NASCOP), MINISTRY OF PUBLIC HEALTH AND SANITATION
1
National AIDS Control Organisation (NACO)
1
National AIDS Control Organisation (NACO), Ministry of Health and Family Welfare, Government of India
1
National AIDS Control Organisation Ministry of Health & Family Welfare, Government of India
1
National AIDS Control Organization (NACO), Ministry of Health and Family Welfare (MOHFW), Government of India
1
National Alliance on Mental Illness
1
National Bureau of Statistics (NBS), National Primary Health Care Development Agency (NPHCDA) and National Agency for the Control of Aids (NACA)
1
National Center for AIDS and STD Control (NCASC)
1
National Center for Disease Prevention and Control Department of Health, Philippines
1
National Center for Emerging and Zoonotic Infectios Diseases
1
National Center for Emerging and Zoonotic Infectious Diseases (NCEZID)
1
National Center for Tuberc ulosis and Leprosy Control (CENAT) , Cambodia
1
National Center for Tuberculosis and Leprosy Control (CENAT)
1
National Center for Tuberculosis and Leprosy Control (CENAT), Cambodia
1
National Centre for AIDS and STD Control (NCASC)
1
National Department of Health (NDoH), Statistics South Africa (Stats SA), South African Medical Research Council (SAMRC), and ICF
1
National Department of Health, South Africa
1
National Dissemination Center for Children with Disabilities (NICHCY)
1
National HIV
1
National Institute of Environmental Health Sciences
1
National Institute of Health, Pakistan
1
National Institute of Mental Health & Neurosciences, Bengaluru India
1
National Institute of Population Research and Training (NIPORT), Mitra and Associates, and ICF International
1
National Institute of Statistics (NIS) [Rwanda], Ministry of Health (MOH) [Rwanda], and Macro International Inc.
1
National Institute of Statistics of Rwanda (NISR) [Rwanda], Ministry of Health (MOH) [Rwanda], and ICF International
1
National Institute of Statistics of Rwanda (NISR) [Rwanda], Ministry of Health (MOH) [Rwanda], and ICF International.
1
National Institutes of Health
1
National LGBTQ Task Force
1
National Libary of Medicine
1
National Library of Medicine
1
National Library of Medicine (NIH)
1
National Malaria Control Programme (NMCP) Communicable Disease Control Division Directorate General of Health Services Ministry of Health & Family Welfare Bangladesh
1
National Malaria Control Program (NMCP) [Liberia], Ministry of Health (MOH), Liberia Institute of Statistics and Geo-Information Services (LISGIS), and ICF
1
National Maternal & Child Oral Health Resource Center
1
National Polio Emergency Operations Centre (EOC), Islamabad, Pakistan
1
National Population Commission (NPC
1
National Primary Health Care Development Agency
1
National Primary Health Care Development Agency, Abuja, Nigeria
1
National Public Health Institute of Liberia (NPHIL)
1
National Rural Health Mission, DOTS
1
National Sexual Violence Resource Center
1
National Social Mobilisation Pillar
1
National Statistical Office Zomba, Malawi
1
National Statistical Office (NSO) and ICF Macro
1
National Statistical Office Zomba, Malawi
1
National TB
1
National Tuberculosis and Leprosy Programme Ministry of Health and Social Welfare, Tanzania
1
National Tuberculosis, Leprosy and Lung Disease Unit, Ministry of Health, Kenya
1
nature communications
1
NCDC
1
NCEZID
1
NCEZID, CDC
1
NCTSN
1
Ndiaye, Salif, et Mohamed Ayad
1
NdOH South Africa
1
NED Foundation nurses volunteers
1
Nepal GHI Strategy
1
Nepal Ministry of Health and Population, New ERA, and ICF International
1
Net Hope
1
New England Journal of Medicine
1
New Jersey Medical School
1
NIH
1
NIOSH
1
Nisha Bhat, Peter H. Kilmarx, Freeman Dube, et al.
1
Nunes da Cunha I., Autry E. et al
1
Nunn, Marc
1
Nữ tu Bác sĩ Nguyễn Thị Hiền
1
NWP, Aqua for All,Kit Publisher et al.
1
O. Chokoshvili
1
O. O. Simooyqa
1
Obiezue, RN.
1
OCHA
1
Odlum, A.
1
OECD
1
Office of Rule of Law and Security Institutions
1
Official Journal of the Asian Pacific Society of Respirology
1
Ogden Stephanie et al
1
Ogden, S., Gallo, K., Davis, S., et al
1
ohns Hopkins Center for Communication Programs
1
OiE
1
Okoye, CI.
1
Olans R. N. , R. D. Olans, A. DeMaria Jr
1
OPAS
1
Open Heart
1
Open Society Institute - Public Health Program
1
OPP-ERA, et al.
1
OPS
1
OPSIC
1
Oranizatión Panamerica de la Salud
1
Organisation de la Santé Mondiale, Bureau Regional de l'Afrique
1
Organisation internationale du Travail (OIT)
1
Organisation mondiale de la Santé (Amériques)
1
Organisation panaméricaine de la Santé
1
Organiuación Mundial de la Salud, Américas
1
Organización Mundial de la Salud (Américas)
1
Organización Mundial de la Salud OMS
1
Organização Mundial da Saúde
1
Organizaición Mundial de la Salud
1
Oritz, G.
1
OSHA
1
Oxfam
1
Ozioko ,KU.
1
O’Neill, J.
1
P. Ndokweni
1
P. De Castro
1
P. Idele
1
P. M. Enright, J. Shumake-Guillemot et al.
1
P. Silva
1
P. Zheng
1
Palmero, D.
1
Pan African Medical Journal
1
Pan American Health Organization (Regional Office of the World Health Organization)
1
Pandey, J. P., M.R. Dhakal, S. Karki, P. Poudel, and M.S. Pradhan
1
Panel on Antiretroviral Guidelines for Adults and Adolescents
1
Panel on Antiretroviral Therapy and Medical Management of HIV-Infected Children
1
Panel on Guidelines for the Prevention and Treatment of Opportunistic Infections in Adults and Adolescents with HIV
1
Panel on Opportunistic Infections in HIV-Infected Adults and Adolescents
1
Patricio V. Marquez, and Shekhar Saxena
1
Paul J. Schramm, Munerah Ahmed, Hannah Siegel, Jamie Donatuto et al.
1
Paul Roddy, Natasha Howard, Maria D. Van Kerkhove, et al.
1
Peace Corps
1
Peer J
1
PERC
1
Petry, Nicolai and others
1
Phillipine Department of Health
1
Phimemon, Rune N., Michael J. Mahande, and Habib O. Ramadhani
1
PLOS journals
1
PLoS Neglected Tropical diseases
1
PMC (US National Library of Medicine National Institutes of Health)
1
Pollack L. A., D. Plachouras, H. Gruhler et al.
1
Population Reference Bureau
1
President's Malaria Initiative
1
President's Malaria Initiative et al.
1
President’s Malaria Initiative, CDC, MCHIP, MCSP
1
Prison Department
1
Prof. AHM E. Hussain
1
Prof. Dr. A. K. Azad
1
Prof. Dr. S. Tahamina
1
Prof. Q. T. Islam
1
Programme National de lutte contre la Tuberculose (PNT) - Sénégal
1
Programme national de lutte contre la tuberculose - Sénégal
1
PROMUNDO-US, UNFPA
1
Puerto Castro GM, Montes Zuluaga FN, Alcalde-Rabanal JE, Pérez F, et al.
1
Quinn, V., and A. Guyon
1
R. and Coimbra Brito
1
R. Baggaley
1
R. Bucciardini
1
R. Doshi
1
R. Elovich
1
R. H. Remien
1
R. James
1
R. Leyva-Flores
1
R. L’Herminez
1
R. Pebody
1
R. Saucier
1
R. Thompson, G. Fontana, et al.
1
R. Velayudhan
1
R. Zahn
1
RACGP
1
Rajiha A. Ibrahim, Amete M. Teshal, Surafel F. Dinku, et al.
1
Ravindranath R Tongaonkar, David L Sanders and Andrew N Kingsnorth
1
RCCE technical working group
1
Rebuilding Basic Health Services Project (RBHS)
1
Rediscovering Biology
1
Rehabilitation Division
1
Republic of Bangladesh, Ministry of Health and Family Welfare
1
Republic of Kenya, Ministry of Health
1
Republic of Libera, Ministry for Health and Social Welfare
1
Republic of Liberia
1
Republic of Rwanda
1
Research Triangle Institute (RTI) International
1
Restless Development
1
Results for Development (R4D)
1
Review on Antimicrobial Resistance
1
Revista Medica Uruguay
1
Richards, P.
1
Robert Koch Institut RKI
1
Robert Wood Johnson Foundation
1
Robin G. and J. Balbus
1
Rockville, Maryland, USA: ICF International
1
Rontgene M. Solante, Manolito L. Chua
1
Rosenbaum, Julia and Bery, Renuka
1
Roy, S. et al.
1
Rudolf,L-
1
Rugigana, Etienne, Francine Birungi, and Manassé Nzayirambaho
1
Rupp, N.
1
Rutstein, S.E., J. Ananworanich, S. Fidler, et al.
1
Rwanda Biomedical Center
1
Rwanda Biomedical Center, National Reference Laboratory Division
1
S. Buchan
1
S. Colt, M.N. Garcia-Casal, J.P. Peña-Rosas, et al.
1
S. Galwankar
1
S. H.-L. Fry
1
S. I. Ngulde
1
S. Ijaz
1
S. Kaba
1
S. L. Barnabas
1
S. Meshnick, M. Janko, S. Doctor, et al.
1
S. Nachman et al.
1
S. Richiedei
1
S. Tassew Seboka, A. Hawkins et al.
1
S.B. Calderwood and A.Bloom
1
SAARC TB and HIV
1
SAARC Tuberculosis and HIV
1
Sachs, J., Karim, S., Aknin, L. et al.
1
Safrani, N.
1
Sage Working Group on Vaccine Hesitancy
1
Saiful Islam M. , Mizanur Rahman K., Sun Y., et al.
1
Sandra F. Moreira-Silva1,3, Eliana Zandonade
1
Santesso, N.
1
Santibañez, S., V. Spiegel, M. O'Sullivan, et al
1
Saori Ohkubo, Tara M. Sullivan, Sarah V. Harlan, Barbara K. Timmons, Molly Strachan
1
Saparbekov M. K.
1
Saranya A, Selvaraj E Lee, Mason Harrell, et al.
1
Save the Children andJohn Hopkins Center for Communication Programs
1
Schramm, P,J.
1
Scientific and Cultural Organization
1
Scott F. Dowell, James J. Sejvar, Lul Riek, et al.
1
Scott, C.
1
Secretaria de Estado de Saúde de Minas Gerais (SES-MG), Brasil
1
Segvia, E. V. L. et al.
1
Sekamatte, M.
1
Seputra, K.P., Tarmono, B.S. Noegroho, et al.,
1
Seung Hee Lee-Kwan, et al.
1
Sharafi, L. et al.
1
Shears, Paul, T.J.D. O'Dempsey
1
Shekhar Saxena , Etienne Krug, Oleg Chestnov et al.
1
Shemsedin Toyba et al.
1
Shoaib Fahad Hussai, Peter Boyle, Preeti Patel and Richard Sullivan
1
SickKids
1
Sightsavers, Department for International Development, The International Trachoma Initiative , et al.
1
SIGN
1
Sigridur B.Thormar
1
SMAC
1
Smithsonian Alerts
1
Social Science in Humanitarian Action
1
Social Science in Humanitarian Action: A Communication for Development Platform
1
Sociedad ecuatoriana de geriatria y gerontologia
1
Sociedad Venezolana de Salud Pública
1
Society of Hospital Medicine
1
Soka, M.J.
1
Solidarités International
1
Solthis, et al.
1
south
1
South Africa's Dept. of Health
1
South to South Zoe life
1
Souto Medeiros, D. et al.
1
Spigel, Lauren
1
Springer Science+Business Media
1
SpringerLink
1
Standleya C. J., Carlinb E. P., Sorrella E. M. et al.
1
Staples JE, Dziuban EJ, Fischer M, et al
1
Statistics Sierra Leone, Ministry of Health and Sanitation Sierra Leone, ICF International
1
Stefan Mauss, et al.
1
Stone, D.
1
Stop TB
1
STRIVE
1
Suicide Prevention Resource Center
1
Susa Coffey,et al.
1
T. Bingham
1
T. Colton
1
T. Fountaine, J. Lembong, R. Nair, et al.
1
T. Frieden
1
T. Johnson, Ph.D., et al.
1
T. Jones and J. Bedford
1
T. Kasrashvili
1
T. Leenstra, MD, PhD
1
T. Leonhardt
1
T. Porth
1
T. Schaberg, T. Bauer, F. Brinkmann, et al.
1
T. T. Bella-Awusah
1
T. Welsh
1
T. Zurashvili
1
Takarinda, Kudakwashe C., Lydia K. Madyira, Mutsa Mhangara, Victor Makaza, Memory Maphosa, Simbarashe Rusakaniko, Peter H. Kilmarx, Tsitsi Mutasa-Apollo, Getrude Ncube, and Anthony D. Harries
1
Tamara Adrián-Hernandez,Elham Atalla, George Ayala et al.
1
Tambo, Ernest
1
Tarun Gera, Dheeraj Shah, Paul Garner et al.
1
TB
1
TB HIV
1
TB online
1
TB/CTA
1
TDR Program
1
Tefera, S.
1
Teferi, M.
1
Teh DHS Program
1
Telligen, Quality Innovation Network National Coordinating Center, Centers for Medicare & Medicaid Services (CMS)
1
The Brookings Institution’s International Volunteering and Service Initiative
1
The Center for Disease Dynamics, Economics & Policy CDDEP
1
The Center for HIV Law Policy
1
The Dag Hammarskjöld Foundation and ReAct
1
The DHS Program ICF
1
The DHS Program ICF International
1
The Estonian Ministry of Justice
1
The Ethiopian Public Health Institute
1
The European Respiratory Journal
1
The Focus Group Consulting
1
The Global Fund
1
The Global Public Private Partnership for Handwashing
1
The Global Virology Therapy Monitor Team, Ipsos Healthcare
1
The Government of Malawi
1
THE GOVERNMENT OF THE KINGDOM OF SWAZILAND MINISTRY OF HEALTH AND SOCIAL WELFARE
1
The Indonesian Association of Pulmonologist
1
The International Center for AIDS Programm
1
The International Federation of Anti-Leprosy Associations (ILEP)
1
The International Federation of Red Cross and Red Crescent Societies IFRC
1
The International Spinal Cord Association
1
The Johns Hopkins Center for Humanitarian Healt
1
The Kenya Mentor Mother Program
1
The Lancet Commissions
1
The Lancet Global Health
1
the Liberia Institute of Statistics and Geo-Information Services (LISGIS) and the University of Liberia Pacific Institute of Research and Evaluation (UL-Pire)
1
The MEDBOX Team
1
The National Academies
1
The National Child Traumatic Stress Network
1
The Partnership Center
1
The Republic of the Union of Myanmar, Ministry of Health and Sports, Department of Health
1
The Republic of the Union of Myanmar, National Disaster Management Committee
1
The Strengthening High Impact Interventions for an AIDS-free Generation (AIDSFree) Project
1
The Tuberculosis Coalition for Technical Assistance (TBCTA)
1
The United States President's Emergency Plan For Aids Relief
1
The United States President's Emergency Plan for AIDS relief (PEPFAR)
1
The World Bank Group
1
Thomson, M.C.
1
Ties Boerma, Colin Mathers, Carla AbouZahr, Somnath Chatterji, Daniel Hogan and Gretchen Stevens
1
Tigrey Health Bureau
1
TK Sundari Ravindran
1
Torres, A.
1
Training NRM School of Medicine
1
Transatlantic Taskforce on Antimicrobial Resistance (TATFAR)
1
Tuberculosis Coalition for Technical Assistance (TBCTA), USAID
1
Tufts University, Oxfam
1
Tylor & Francis
1
U.S Department of health and human service
1
U.S. Department of Health & Human Services
1
U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES, Centers for Disease Control and Prevention
1
U.S. Department of Health and Human Services, Agency for Toxic Substances and Disease Registry (ATSDR)
1
U.S. Department of Health and Human Services. Centers for Disease Control and Prevention, National Center for Emerging and Zoonotic Infectious Diseases
1
U.S. Global Change Research Program
1
U.S. Global Malaria Coordinator
1
U.S. Government
1
U.S. President's Emergency Plan for AIDS Relief
1
U.S. Presidents Malaria Initiative
1
U.S. President’s Malaria Initiative (PMI), CDC, USAID, et al.
1
Uganda Aids Commission, Republic of Uganda
1
UN Children´s Fund
1
UN Mission for Ebola Emergency Response
1
UN Office for the Coordination of Humanitarian Affairs
1
UN Office for the Coordination of Humanitarian Affairs, Government of Madagascar, UN Country Team in Madagascar
1
UN RC Malawi
1
UNAIDS (Joint United Nations Programme on HIV
1
UNAIDS (Joint United Nations Programme on HIV/AIDS)
1
UNAIDS, The National AIDS Authority (NAA)
1
UNAIDS/WHO Working group on global HIV/AIDS and STI surveillance
1
UNCT Malawi
1
UNDP (United Nations Development Programme)
1
UNDP, IRGT, UNFPA, UNAIDS, WHO, USAID, PEPFAR, UCSF Center of Excellence for Transgender Health, Johns Hopkins Bloomberg School of Public Health
1
UNFPA et al.
1
Unicef (Eastern and Southern Africa Office)
1
Unicef (United Nations Children’s Fund)
1
UNICEF Indonesia
1
UNICEF Supply Division
1
Unicef, et al.
1
UNICF
1
Unitaid
1
UNITE FOR DIABETES PHILIPPINES
1
United National Children’s Fund (UNICEF)
1
United Nations Department of Peace Operations
1
United Nations Educational
1
United Nations Office on Drug and Crime UNODC
1
United Nations office on drugs and crime (Vienna)
1
United Nations Office4 on Drugs and Crime
1
United Nations Population Fund
1
United Republic of Tanzania, Ministry of Health and Social Welfare
1
United States Government Zambia Interagency Team
1
United States of America - Department of State
1
Univ. Zagreb
1
University of Buffalo
1
University of Cambridge
1
University of Kwa-Zulu Natal
1
University of Nairobi
1
University of Washington
1
University of Zambia
1
University of Zambia, et al.
1
Universtität Innsbruck
1
UNODC (United Nations of Drugs and Crime)
1
US Agency for International Development, Centers for Disease Control and Prevention
1
US Department of Homeland Security, US Department of Health and Human Services
1
US Dept.Health
1
US Embassy Freetown
1
USAID (From the American People)
1
USAID (U.S. President’s Emergency Plan for AIDS Relief)
1
USAID Deliver Project, Task Order
1
USAID From the American People
1
USAID, SPS, Strengthening Pharmaceutical Systems Program-Kenya
1
USAID, CDC
1
USAID, CRS
1
USAID, et al.
1
USAID, GHKC
1
USAID, SPRING
1
USAID, StopTBPartnership
1
USAID, Strengthening Pharmaceutical Systems SPS
1
V. Allan
1
V. Allread
1
V. Fragola
1
V. Habiyambere
1
V. Krishnasamy, L.Bulage et al.
1
V. M. Kuehlkamp
1
V. Parikh
1
V. Pathak, Z. Harrington, C. C. Dobler
1
V. Tucci
1
Vaidyanathan, A.
1
Valerie Karr et al.
1
van den Dool C., PhD
1
Velho Barreto de Araújo, T.
1
Verbeek, J.H.
1
Verjeen, M.
1
Viện Vệ sinh dịch tễ Trung ương
1
Vikram Patel, Dan Chisholm, Tarun Dua et al.
1
Villella, Christina
1
W. Haas
1
Wakefield, M.K.
1
Walker, N.F.
1
Wambugu, Samuel
1
WASH Cluster
1
Wasif Malik, M.
1
Waterfeld, K.C.
1
Weber, Stephanie
1
Weiss, A.
1
White, Z.
1
WHO Regional Office for Europe
1
WHO, Department of Reproductive Health and Research, London School of Hygiene and Tropical Medicine, South African Medical Research Council
1
WHO, HHS-CDC, GAP
1
WHO, Unicef
1
Wilkinson, S.
1
Willy McCourt, W. & Awases, M.
1
Wirth, James P.
1
Wood MC, DePaolo, F, Whitaker RD
1
Woodhead M., F. Blasi, S. Ewig, et al.
1
World Allergy Organization-IAACI (WAO)
1
World Bank, et al.
1
World Diabetes Foundation
1
World Economic Forum et al.
1
World Food Programme
1
World Health Organization (WHO), Regional Office for South-East Asia
1
World Health Organisation
1
World Health Organisation (WHO) Regional Office for Africa
1
World Health Organisation WHO
1
World Health Organisation, International Labour Organization
1
World Health Organization (Africa)
1
World Health Organization (Bangladesh)
1
World Health Organization (Regional Office for South-East-Asia)
1
World Health Organization (Western Pacific Region)
1
World Health Organization (WHO) Regional Office for the Eastern Mediterranean
1
World Health Organization (WHO), Inter-Agency Task Team (IATT), UNICEF
1
World Health Organization (WHO)
1
World Health Organization et al.
1
World Health Organization Regional
1
World Health Organization WHO (Africa)
1
World Health Organization WHO, Regional Office for Europe
1
World Health Organization WHO, Bangladesh
1
World Health Organization WHO, CDC, UNHCR
1
World Health Organization WHO, Regional Office for Africa
1
World Health Organization WHO, Regional Office for Western Pacific
1
World Health Organization WHO, Unicef
1
World Health Organization, CDC, GTSS et al.
1
World Health Organization, et al.
1
World Health Organization, IASC
1
World Health Organization, Regional Office Africa
1
World Health Organization, Sout-Easr Asia
1
World Health Organization, South-East Asia
1
World Health Organization, TB HIV working group, Stop TB Partnership
1
World Health Organization, UN Office for the Coordination of Humanitarian Affairs, UN Mission for Ebola Emergency Response
1
World Health Organization,et al.
1
Worldometer
1
Xiaoxu Wu, Yongmei Lu, Sen Zhou et al.
1
Y .V. Kruglov
1
Y. Tabi
1
Y.-H. Jin
1
Yale, G.
1
Yelizaryeva A. V.
1
Z. T. Gavry
1
Z.-S. Cheng
1
Zachary, Blake, Zhuzhi Moore, and Pav Govindasamy
1
Zaidel, E.J.
1
Zambia Ministry of Health, National Food and Nutrition Commission and Food and Nutrition Technical Assistance III Project (FANTA)
1
Zambia National Public Health Institute
1
Zimbabwe National Statistics Agency (Z IMSTAT) and ICF International. 20
1
Zimbabwe National Statistics Agency (ZIMSTAT)
1
Всемирная организация здравоохранения (ВОЗ)
1
Всемирная организация здравоохранения (Европа)
1
Департамент "Остановить туберкулез", ВОЗ
1
Департамент по ВИЧ/СПИД, Департамент «Остановить ТБ», Всемирная организация здравоохранения
1
Дженнифер Батлер
1
Джорджа Айалы
1
Камкин Евгений Геннадьевич, Костенко Наталья Алексеевна et. al.
1
С. Н. Авдеев , Е. В. Волчкова , О. М. Драпкина, et al.
1
Фонд Организации Объединенных Наций в области народонаселения
1
Publication Years
2020-2025
446
2010-2019
1270
2000-2009
142
1990-1999
8
Category
Countries
599
Clinical Guidelines
185
Public Health
103
Key Resources
85
Women & Child Health
63
Pharmacy & Technologies
26
Capacity Building
25
Toolboxes
COVID-19
204
HIV
174
Ebola
168
TB
152
Rapid Response
115
AMR
96
Zika
60
NCDs
53
NTDs
52
Mental Health
41
Planetary Health
34
Cholera
32
Specific Hazards
28
Caregiver
28
Polio
25
Natural Hazards
21
Refugee
21
Pharmacy
15
Global Health Education
11
Disability
9
Conflict
7
Health Financing Toolbox
5
Social Ethics
1
The Namibia Population-based HIV Impact Assessment (NAMPHIA) 2017 | The Ministry of Health and Services is leading the NAMPHIA survey in collaboration with the Namibia Statistics Agency (NSA)
...
and the Namibia Institute of Pathology (NIP). The survey is supported by the United States President’s Emergency Plan for AIDS Relief (PEPFAR), through the U.S. Centers for Disease Control and Prevention (CDC). | The goal of NAMPHIA is to examine the current distribution of the HIV epidemic and assess the impact of Namibia’s prevention, care and treatment response across all 14 regions of Namibia.
more
Belgian Antibiotic Policy Coordination Committee Policy paper for the 2014-2019 term
Erica Balligand, Michiel Costers and Evelyne Van Gastel
Belgian Antibiotic Policy Coordination Committee
(2014)
C2
Antimicrobial resistance represents a big threat to public health. The Centers for Disease Control and
...
Prevention (CDC) estimate that every year two million Americans are infected with a (multi-)drug resistant bacterium, resulting in 23,000 deaths. The WHO has repeatedly drawn attention to this major health issue. In the worst-case scenario, we will shortly run out of effective antibiotics. Surgery and cancer therapy will then become very dangerous due to the risk of infection associated with such treatments. (Organ) transplantation will become close to impossible as the immunosuppression necessary for transplant patients makes them highly vulnerable to infections. Some infections we can easily treat today could turn deadly. It is therefore conceivable that infectious diseases once again become the leading cause of death as in early 20th century.
more
Ethiopia GATS was implemented by Ethiopia Public Health Institute (EPHI) in collaboration with the Ethiopian Food, Medicine, Health Care Administration and Control Auth
...
ority (FMHACA), CSA, FMOH and the World Health Organization (WHO)country office. Technical assistance for the implementation of the survey was provided by the WHO, the U.S. Centers for Disease Control and Prevention (CDC), and RTI International. Program support was provided by the CDC Foundation.Financial support for Ethiopia GATS was provided by the CDC Foundation with a grant from the Bill & Melinda Gates Foundation.GATS enhances countries’ capacity to design, implement and evaluate tobacco control programs. It also assistscountries to fulfill their obligations under the WHO FCTC to generate comparable data within and across countries. In addition,it allows countries to implement the WHO MPOWER policy package. WHO MPOWERisa technical packagedevelopedtoassist countries in implementing selected demand reduction measures contained in the WHO Framework Convention on Tobacco Control(FCTC)(5).The six MPOWER evidence-based measures contained in the FCTC;
more
Version 1.1. The WHO protocol has been adapted to resource-limited settings and builds on existing methodologies from the European Centre for Disease Prev
...
ention and Control (ECDC), the Global PPS project from University of Antwerp, the US Centers for Disease Control and Prevention (CDC), and the Medicines Utilisation Research in Africa (MURIA).
Point Prevalence Surveys collects information on prescribing practices of antibiotics and other information relevant to treatment and management of infectious diseases in hospitalized patients, and complements surveillance of antimicrobial consumption.
more
The article "Non-communicable diseases, injuries, and mental ill-health in Africa: the role of the Africa Centres for Disease
...
Control and Prevention" examines the role of the Africa CDC in addressing non-communicable diseases (NCDs), injuries, and mental health across the continent. It highlights how Africa CDC, originally focused on communicable disease response, has expanded its mandate to include NCDs and mental health, driven by the high mortality and morbidity associated with these conditions.
more
Afin de vous protéger, suivez ces étapes pour retirer des gants
The goal of this addendum is to help management and staff
minimize the risk of TB transmission at facilities in resource limited settings in whi
...
ch a.) HIV-infected persons receive diagnosis, care, treatment,
and/or support, and b.) there is a high prevalence of HIV infection, both known
and undiagnosed, in settings such as prisons, jails, other
detention centers, and drug rehabilitation centers.
more
Africa CDC Non Communicable Diseases, Injuries Prevention and Control and Mental Health Promotion Strategy
recommended
Non-communicable diseases (NCDs) & injuries and mental health conditions constitute a serious impediment to achieving the vision of Agenda 2063 to build an integrated, prosperous,
...
and peaceful Africa driven by its own citizens. Each year, these conditions cause millions of premature deaths and disabled lives across Africa. These conditions also lead to annual economic loss of multiple billion US-Dollars. Their burden both in terms of disease morbidity/mortality and socio-economic impact is increasing.
more
Training manual
This statement aims to provide guidance and recommendation to African Union Member States in responding to the following questions:
What should countries do when announcing identification of a
...
herbal remedy or medicine?
What are the implications for the rest of the continent?
more
24 September 2021. The vaccine has been authorized by the Chinese National Regulatory Authority (NRA) – the National Medicinal Product Administration (NMPA) – as well as other regulatory authorities. Because WHO has granted Emergency Use Listing (EUL) for the Sinovac COVID-19 vaccine (Vero Cell
...
[Inactivated]) – CoronaVac, the Africa Union and Africa CDC’s Regulatory Taskforce has endorsed the Emergency Use Authorisation for the vaccine (see scenario #1). As such, African Union Member States are recommended to waive any review processes and rely directly on the WHO EUL via the AVAREF managed pathway described in Scenario #1
more
The Sierra Leone National Infection Prevention and Control Guidelines were jointly developed and
...
updated by the Ministry of Health and Sanitation in collaboration with the World Health Organization and the US Centers for Disease Control and Prevention.
more
Morbidity and Mortality Weekly Report (MMWR) January 16, 2015 / 64(01);20-27