Filter
1108
Text search:
molecular
testing
Featured
154
232
Language
Document type
539
235
180
57
47
24
12
9
2
2
1
Countries / Regions
70
41
36
27
26
24
23
20
18
17
13
13
13
11
11
11
10
9
9
8
8
8
8
8
8
7
7
7
7
6
6
6
5
5
5
5
5
5
5
5
5
4
4
4
4
4
3
3
3
3
3
3
3
3
3
2
2
2
2
2
2
2
2
2
2
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
Authors & Publishers
Publication Years
Category
328
162
52
43
21
14
4
Toolboxes
250
154
81
75
60
58
32
25
23
20
19
13
12
12
8
6
5
4
4
3
3
2
WHO needs US$2.54 billion to provide life-saving assistance to millions of people around the world facing health emergencies. WHO’s Health Emergency Appeal is a consolidation of WHO’s priorities and financial requirements for 2023 to carry out health interventions in emergency and humanitarian r
...
Report of the WHO/Bill & Melinda Gates Foundation Consultation. The Consultation was organized back-to-back with the first annual meeting of the International Coordinating Group of the BMGF-funded project for human and dog rabies elimination in developing countries, held at WHO headquarters, Geneva,
...
In 2005, the World Health Organization (WHO) recognized Chagas disease (CD; Trypanosoma cruzi infection) as a neglected tropical disease (NTD) [1] and included it into the global plan to combat NTDs [2]. The Target 3.3 of the United Nations Sustainable Development Goals (UN/SDG) aims at ending the e
...
BMJ Glob Health 2017;2:e000345. doi:10.1136/bmjgh-2017-000345. WHO's 2020 milestones for Chagas disease include having all endemic Latin American countries certified with no intradomiciliary Trypanosoma cruzi transmission, and infected patients under care. Evaluating the variation in historical expo
...
This guideline for the prevention and control of chikungunya fever
(CF) is intended for use by all peripheral health workers in the Region and
is based on the strategy outlined above. This document will focus mainly
on preventing, predicting and detecting outbreaks, and after detection,
investig
...
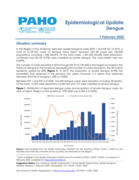
Epidemiological Update
Dengue
7 February 2020
Situation summary
In the Region of the Americas, between epidemiological week (EW) 1 and EW 521 of 2019, a total of 3,139,335 cases of dengue have been reported (321.58 cases per 100,000 population), including 1,538 deaths. Of the total cases, 1,367,
...
Arsenical monotherapies were previously very successful for treating human African trypanosomiasis (HAT).
Melarsoprol resistance emerged as early as the 1970s and was widespread by the late 1990s.
Melarsoprol resistance represents the only example of widespread drug resistance in HAT patients wher
...
Full Perscribing information on Fexinidazole Tablet for oral use
INDICATIONS AND USAGE
Fexinidazole Tablets are indicated for the treatment of both the first-stage (hemolymphatic) and second-stage (meningoencephalitic) human African trypanosomiasis (HAT) due to Trypanosoma brucei gambiense in pati
...
In support of the London Declaration goals, PATH aims to catalyze engagement of the diagnostics industry and product development efforts. As part of this work, PATH conducted a diagnostic landscape analysis to identify gaps and evaluated current and nascent HAT diagnostics that may provide solutions
...
The development of this target product profile (TPP) was led by the WHO Department of Control of Ne-
glected Tropical Diseases (NTD) following standard WHO guidance for TPP development. In order to
identify and prioritize diagnostic needs, a WHO NTD Diagnostics Technical Advisory Group (DTAG)
was
...
This handbook is intended primarily for front-line health care providers who are likely to see children (among other clients) in their day-to-day practice. These may include general practitioners, nurses, midwives, gynaecologists,
paediatricians, mental health professionals, first responders and st
...
Research and Reports in Tropical Medicine 2022:13 25–40.
Chagas disease (CD) is caused by the parasite Trypanosoma cruzi, and it is endemic in Central, South America, Mexico and the South of the United States. It is an important cause of early mortality and morbidity, and it is associated with po
...
This technical report presents the epidemiology of human and animal leishmaniases in the EU and its neighbouring countries and concludes that the disease remains widespread and underreported in many countries of southern Europe, northern Africa, and the Middle East and that there is a need to improv
...
In response to the recent publication “Is onchocerciasis elimination in Africa feasible by 2025: a perspective based on lessons learnt from the African control programmes” by Dadzie et al., it is important to clarify and highlight the positive and unequivocal research and operational contributio
...
Leptospirosis is a bacterial zoonotic disease of worldwide importance, though relatively neglected in many African countries including sub Saharan Africa that is among areas with high burden of this disease. The disease is often mistaken for other febrile illnesses such as dengue, malaria, rickettsi
...
Leptospirosis, a spirochaetal zoonosis, occurs in diverse epidemiological settings and affects vulnerable populations, such as rural subsistence farmers and urban slum dwellers. Although leptospirosis is a life-threatening disease and recognized as an important cause of pulmonary haemorrhage syndrom
...
This analysis focused on the chronic form of HAT caused by T. b. gambiense, as it contributes to the majority of disease burden. Information from the literature review,
product development landscape, and stakeholder interviews was compiled to:
- Identify use cases and understand current diagnosti
...
The objectives of the meeting were:
1. To step up the commitment of national authorities and technical and financial partners toWHO’s elimination objective for g-HAT.
2. To share achievements, challenges and views on the elimination goal among countries and implementing partners.
3. To assess t
...
Taeniasis and cysticercosis caused by the parasite T. solium affect vulnerable populations,
mainly in Latin America, sub-Saharan Africa and Asia, where pigs (the intermediate host) roam
free and poor sanitation allows pigs access to human faeces.
Rabies is entirely preventable, and vaccines, medicines, tools and technologies have long been available to prevent people from dying of dog-mediated rabies. Nevertheless, rabies still kills about 60 000 people a year, of whom over 40% are children under 15, mainly in rural areas of economically dis
...